Clinical
Psychotropic Medication Monitoring in Skilled Nursing: What Your Data Needs to Show

What Is Psychotropic Medication Monitoring in Skilled Nursing?
Psychotropic medication monitoring is the ongoing clinical process of tracking antipsychotic, antidepressant, anxiolytic, and hypnotic drug use across your resident population — reviewing whether each medication is clinically indicated, documented with appropriate diagnoses, and subject to regular reduction attempts where required. CMS publicly reports antipsychotic use rates for every nursing home in the country, and the data feeds directly into Five-Star quality measure scores.
Every quarter, CMS updates a number on Care Compare that families, discharge planners, and managed care organizations look at: the percentage of long-stay residents receiving antipsychotic medications without a qualifying diagnosis.
It's a quality measure. It's public. And for multi-facility operators, it can vary dramatically across buildings — not because of differences in the resident population, but because of differences in how consistently each building is tracking, reviewing, and documenting psychotropic medication use.
Psychotropic medications — antipsychotics, antidepressants, anxiolytics, hypnotics — are among the most closely regulated drug categories in skilled nursing. CMS requires quarterly medication reviews, gradual dose reduction (GDR) attempts for antipsychotics, documented diagnoses for every psychotropic prescribed, and physician sign-off on any refusal of GDR. Surveyors look at it. Families ask about it. And the data lives scattered across physician orders, MDS sections, clinical notes, and pharmacy reports.
For a regional director or VP of Clinical managing 10 or 15 buildings, the only way to stay ahead of a psychotropic monitoring issue is to see the data across the organization — before survey, before a family complaint, before a quality measure drop.
This post covers what CMS tracks, what your data should be showing you, and how multi-facility operators build monitoring systems that work.
What CMS Tracks and Why It Matters for Your Five-Star Score
CMS measures antipsychotic use as part of the Five-Star Quality Measures rating. It appears on Care Compare as a percentage — the share of your long-stay residents receiving antipsychotic medications — and is compared against state and national averages.
The measure excludes residents with a qualifying psychiatric diagnosis (schizophrenia, Tourette's syndrome, Huntington's disease). All other long-stay residents receiving antipsychotics count toward the public measure. The CMS National Partnership to Improve Dementia Care has driven measurable reductions in antipsychotic use nationally since 2012 — but rates remain inconsistent across operators and markets.
| CMS Quality Measure | What It Captures | Where It Appears |
|---|---|---|
| Antipsychotic use (long-stay) | % of residents receiving antipsychotics without qualifying diagnosis | Care Compare public measure |
| Antipsychotic use (short-stay) | % of short-stay residents receiving new antipsychotic prescriptions | Care Compare public measure |
| MDS Section N | Psychotropic medication use recorded per resident | Used for quality measure calculation |
A high antipsychotic use rate affects your quality measure star score — one of the three components of the Five-Star composite. It also signals to surveyors that your facility may need a closer look at medication management practices.
Beyond the rating, inappropriate antipsychotic use carries direct regulatory risk. Unnecessary psychotropic medications are an F-Tag citation category (F758 for unnecessary drugs, F759 for antipsychotics specifically). A citation in this area during an annual survey can result in deficiency findings that affect your health inspection star as well.
The 4 Psychotropic Categories Your Clinical Team Needs to Track
Antipsychotics get the most CMS attention, but psychotropic monitoring in LTC extends across four drug categories — each with its own regulatory requirements and clinical risks.
| Drug Class | Examples | Key Monitoring Requirements |
|---|---|---|
| Antipsychotics | Haloperidol, quetiapine, risperidone | Qualifying diagnosis required; quarterly GDR attempt unless clinically contraindicated; physician sign-off if refused |
| Antidepressants | Sertraline, mirtazapine, escitalopram | Indication documented; regular effectiveness review |
| Anxiolytics / Benzodiazepines | Lorazepam, clonazepam, diazepam | Short-term use expectations; tapering plan where applicable |
| Hypnotics | Zolpidem, temazepam | Duration and indication reviewed; fall risk assessment |
CMS surveyors can and do review all four categories. The antipsychotic measure is publicly reported; the others are assessed during inspection. A facility that tracks antipsychotics closely but doesn't monitor benzodiazepine use across the building creates a regulatory blind spot. The Alzheimer's Association notes that antipsychotics are frequently prescribed to nursing home residents with dementia for behavioral symptoms — a use that is off-label, carries significant safety risks, and is subject to heightened CMS scrutiny.
Where Psychotropic Monitoring Breaks Down in Multi-Facility Operations
The clinical intent behind psychotropic monitoring is clear. The operational challenge is execution across multiple buildings.
Inconsistent Documentation of Diagnoses
CMS requires that every psychotropic prescription be supported by a documented clinical indication. In practice, prescriptions sometimes carry over from prior settings — a resident admitted from a hospital with an antipsychotic on the medication reconciliation list, where the original indication isn't clearly documented in the SNF chart.
When this happens at one building, a DON can catch it during a medication review. When it happens across 12 buildings, only a data system that surfaces residents with active psychotropic orders and missing or unclear diagnoses will catch it consistently.
GDR Tracking Without a Cross-Facility View
Gradual dose reduction attempts for antipsychotics are required quarterly. Each attempt — whether successful, unsuccessful, or declined by the resident or physician — must be documented. If a building misses a GDR cycle because the clinical schedule got disrupted or a new MDS coordinator didn't have the process, the documentation gap may not surface until survey.
For a regional director, knowing which buildings are current on GDR cycles and which have open attempts due requires either a manual audit of each building's pharmacy or medication records — or a clinical dashboard that tracks it in one place.
MDS Section N Accuracy
MDS Section N captures psychotropic medication use for quality measure purposes. If a resident is receiving a psychotropic medication and it's not correctly recorded in Section N, the building's public quality measure won't reflect reality. That can go in either direction: a lower rate that doesn't reflect actual prescribing, or data that mismatches what the pharmacy shows — both of which create survey risk.
New Admissions Risk
Short-stay residents admitted with existing psychotropic prescriptions represent a distinct monitoring challenge. CMS tracks new antipsychotic prescriptions among short-stay residents as a separate quality measure. Facilities with high short-stay volume and active admission pipelines need a process for flagging new psychotropic prescriptions at admission and initiating the appropriate documentation and review cycle immediately.
What Your Data Should Be Showing You
A functional psychotropic monitoring system for multi-facility operators answers these questions, at any time, across every building:
| Question | Why It Matters |
|---|---|
| Which residents are currently receiving antipsychotics? | Baseline census; required for GDR scheduling and rate monitoring |
| Which have a qualifying diagnosis on file? | Missing diagnosis = F-Tag risk and quality measure impact |
| Which are due for a GDR attempt this quarter? | Missed GDR cycle = documentation deficiency |
| Which GDR attempts were refused? | Physician/resident refusal must be documented; not tracked = gap |
| How does each building's antipsychotic rate compare to state average? | Early warning before Care Compare update reflects it |
| Are any new psychotropic prescriptions issued this week? | Short-stay metric and new admission monitoring |
| What is the trend over the past 4 quarters? | Sustained rate increase warrants clinical intervention |
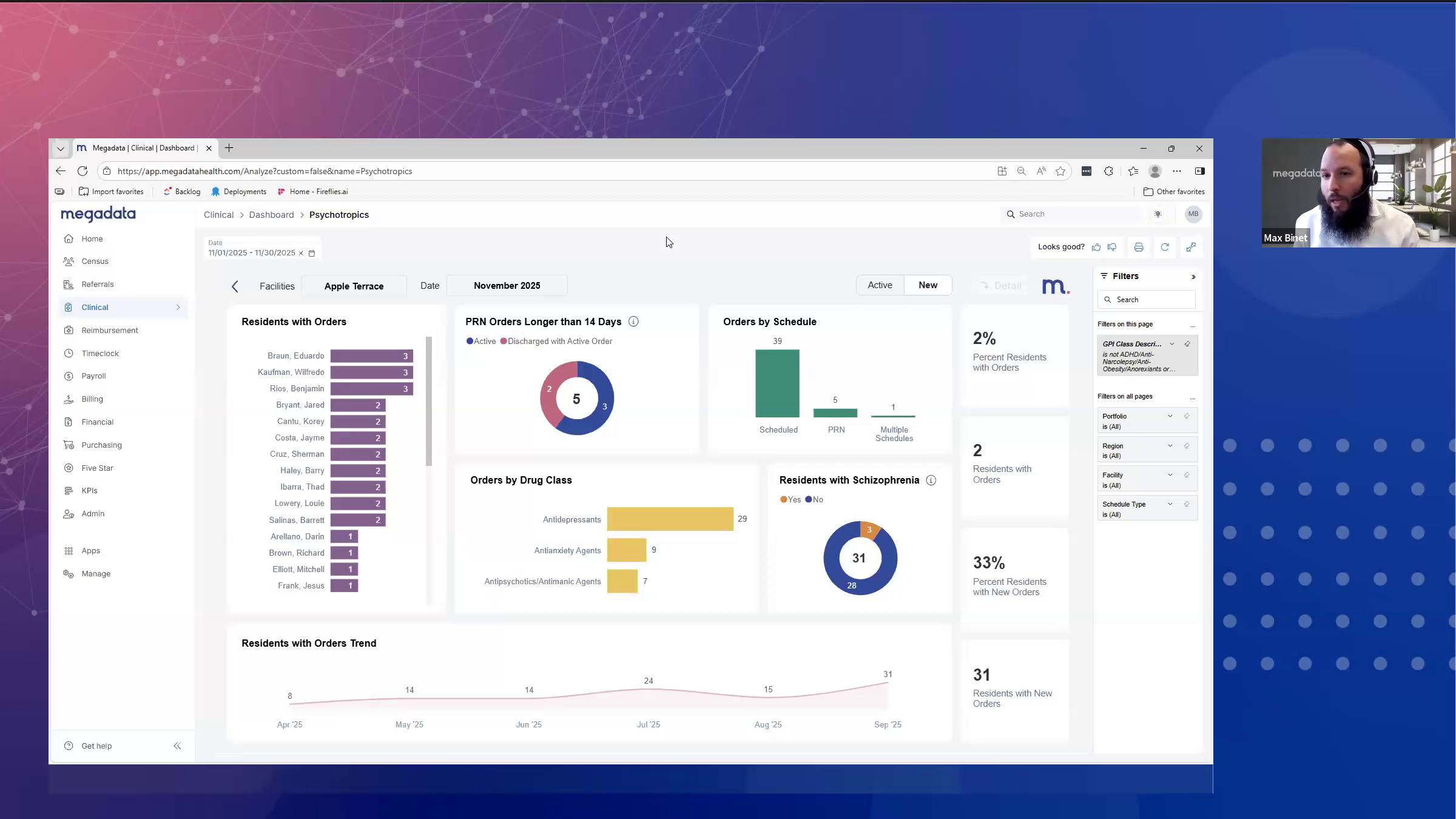
Most of this information lives in a combination of the pharmacy system, EMR, and the MDS. Getting it into one view — across all buildings, updated automatically — is the operational challenge. Megadata's clinical module contains a dedicated psychotropics dashboard that surfaces psychotropic indicators across your organization, so a VP of Clinical can see the rate by building, the GDR cycle status by resident, and the trend over time without pulling reports from each site individually.
{{product-demo-call="/components"}}
Building a Psychotropic Monitoring Workflow That Scales
For operators managing five buildings, a manual monthly review might be enough — barely. For operators managing 15 or 25, it breaks down. Here's the cadence that works at scale:
- New psychotropic prescriptions issued across all buildings (flag for documentation review)
- Any resident with active antipsychotic order and no qualifying diagnosis on file
- Upcoming GDR attempts due in the next 30 days
- Building-level antipsychotic rate compared to state benchmark
- GDR completion status for the quarter to date
- Benzodiazepine and hypnotic use review by building
- MDS Section N audit — do pharmacy records align with what's recorded?
- GDR attempt documentation complete for all applicable residents
- New antipsychotic rate review for short-stay census
- Full psychotropic medication census by building
- Documentation completeness audit for every active psychotropic prescription
- Identify any residents where GDR was refused — verify physician sign-off is on file
The facilities that perform best in survey on medication management aren't doing more than this. They're doing exactly this, consistently, because the data is in front of them — not buried in individual building reports they have to request.
Frequently Asked Questions
What is the CMS antipsychotic quality measure for nursing homes?
CMS publicly reports the percentage of long-stay nursing home residents receiving antipsychotic medications as a Five-Star quality measure. Residents with qualifying psychiatric diagnoses (schizophrenia, Tourette's, Huntington's disease) are excluded. Facilities with rates above the state or national average may see quality measure star score reductions and increased survey scrutiny.
What is gradual dose reduction (GDR) for antipsychotics in SNFs?
GDR is a CMS-required process for residents receiving antipsychotic medications in skilled nursing facilities. Facilities must attempt to gradually reduce the antipsychotic dose quarterly to determine whether the medication is still clinically necessary. If the resident or physician declines, the refusal must be documented. Missing a GDR cycle — or failing to document it — is a deficiency finding under F-Tag F758/F759. AMDA – The Society for Post-Acute and Long-Term Care Medicine publishes clinical practice guidelines on GDR protocols and antipsychotic use in post-acute and long-term care settings.
Which psychotropic medications are regulated in nursing homes?
CMS oversight covers antipsychotics, antidepressants, anxiolytics (including benzodiazepines), and hypnotics. Antipsychotics receive the most regulatory attention and are publicly reported. Anxiolytics and hypnotics are reviewed during annual surveys and can result in F-Tag citations if prescribed without appropriate indication or duration documentation.
How does antipsychotic use affect Five-Star ratings?
CMS includes antipsychotic use rates in the quality measures component of the Five-Star rating. A high rate — especially compared to state or national averages — can lower a facility's quality measures star score, which contributes to the overall Five-Star composite. The antipsychotic measure also appears directly on Care Compare, visible to families, referral sources, and managed care organizations.
How do multi-facility operators track psychotropic monitoring across buildings?
Effectively, it requires a clinical data platform that aggregates medication records, MDS Section N data, and GDR documentation across all buildings in one view. Manual approaches — requesting monthly reports from each building's DON or pharmacy — don't scale past five or six buildings and create inconsistency. Real-time clinical dashboards allow a VP of Clinical to see the antipsychotic rate, GDR status, and documentation completeness across the entire organization at any point. AHCA/NCAL tracks antipsychotic use benchmarks across member facilities as part of its Quality Initiative — providing state and national comparisons operators can use to contextualize their own performance.
What F-Tags are associated with psychotropic medication deficiencies?
F-Tag F758 covers unnecessary drug use — medications without a documented clinical indication, monitoring plan, or appropriate use. F-Tag F759 specifically addresses antipsychotic medications, including requirements for qualifying diagnoses and GDR documentation. Both are cited during annual surveys and can contribute to health inspection deficiency scores, affecting the health inspection component of Five-Star.
Clinical Data That Protects Residents and Quality Scores
Psychotropic medication monitoring isn't primarily a regulatory exercise. The clinical intent — ensuring that every medication a resident receives is appropriate, documented, and regularly reviewed — is the point. The quality measures and survey requirements exist because that level of oversight matters for resident safety and dignity.
For multi-facility operators, the challenge is scale. A DON at a single building can track their 90 residents' psychotropic regimens manually. A VP of Clinical overseeing 15 buildings cannot. Without a cross-facility view, the organization is always reacting — to a survey finding, a quality measure drop, a family concern — rather than staying ahead of it.
The goal is visibility before something becomes a problem. That means seeing the data across your organization, weekly, in one place.
See how Megadata's clinical dashboard tracks psychotropic indicators across your buildings, or book a 20-minute walkthrough to see it in action.